Chemicals
-
 KAIST Develops Novel Catalyst With 100-Fold Platinum Efficiency
Propylene, a key building block used in producing plastics, textiles, automotive components, and electronics, is essential to the petrochemical industry. A KAIST research team has developed a novel catalyst that dramatically enhances the efficiency of propylene production while significantly reducing costs.
< Photo. Professor Minkee Choi (left), and Ph.D. Candidate Susung Lee (right) of the Department of Chemical and Biomolecular Engineering >
KAIST (represented by President Kwang-Hyung Lee) announced on the 12th of May that a research group led by Professor Minkee Choi from the Department of Chemical and Biomolecular Engineering has successfully developed a new catalyst using inexpensive metals—gallium (Ga) and alumina (Al₂O₃)—with only a trace amount of platinum (100 ppm, or 0.01%). Remarkably, this new catalyst outperforms conventional industrial catalysts that use high concentrations of platinum (10,000 ppm).
Propylene is commonly produced through the propane dehydrogenation (PDH) process, which removes hydrogen from propane. Platinum has long been used as a catalyst in PDH due to its high efficiency in breaking carbon-hydrogen bonds and facilitating hydrogen removal. However, platinum is costly and suffers from performance degradation over repeated use.
To address this, the KAIST team engineered a catalyst that incorporates only a minimal amount of platinum, relying on gallium and alumina as the primary components.
< Figure 1. Schematic diagram showing the catalytic cooperation between gallium (Ga) and platinum (Pt) >
The core mechanism of the catalyst involves a cooperative function between the metals: gallium activates the C–H bond in propane to produce propylene, while platinum bonds the residual hydrogen atoms on the surface to form hydrogen gas (H₂), which is then released. This division of roles allows for high catalytic efficiency despite the drastic reduction in platinum content.
The researchers identified an optimal platinum-to-gallium ratio that delivered peak performance and provided a scientific rationale and quantitative metrics to predict this ideal composition.
Additionally, the team addressed a major limitation of traditional platinum catalysts: sintering—the agglomeration of platinum particles during repeated use, which causes performance loss. By adding a small amount of cerium (Ce), the researchers successfully suppressed this aggregation. As a result, the new catalyst maintained stable performance even after more than 20 reaction-regeneration cycles.
< Figure 2. Performance comparison of KAIST's newly developed catalyst (100 ppm platinum) and existing commercial platinum catalyst (10,000 ppm platinum) >
Professor Choi stated, “This research demonstrates the possibility of reducing platinum usage to 1/100th of current levels without compromising, and even enhancing, performance. It presents significant economic and environmental advantages, including lower catalyst costs, extended replacement intervals, and reduced catalyst waste.”
He added, “We are planning to evaluate this technology for large-scale process demonstration and commercialization. If adopted in industry, it could greatly improve the economic viability and efficiency of propylene production.”
The study was led by Professor Minkee Choi as corresponding author, with Ph.D. candidate Susung Lee as the first author. The findings were published in the Journal of the American Chemical Society (JACS), a leading journal in chemistry and chemical engineering, on February 13.※ Paper title: Ideal Bifunctional Catalysis for Propane Dehydrogenation over Pt-Promoted Gallia-Alumina and Minimized Use of Precious Elements※ DOI: https://pubs.acs.org/doi/10.1021/jacs.4c13787
The research was supported by the National Research Foundation of Korea and Hanwha Solutions Corporation.
2025.05.12 View 66
KAIST Develops Novel Catalyst With 100-Fold Platinum Efficiency
Propylene, a key building block used in producing plastics, textiles, automotive components, and electronics, is essential to the petrochemical industry. A KAIST research team has developed a novel catalyst that dramatically enhances the efficiency of propylene production while significantly reducing costs.
< Photo. Professor Minkee Choi (left), and Ph.D. Candidate Susung Lee (right) of the Department of Chemical and Biomolecular Engineering >
KAIST (represented by President Kwang-Hyung Lee) announced on the 12th of May that a research group led by Professor Minkee Choi from the Department of Chemical and Biomolecular Engineering has successfully developed a new catalyst using inexpensive metals—gallium (Ga) and alumina (Al₂O₃)—with only a trace amount of platinum (100 ppm, or 0.01%). Remarkably, this new catalyst outperforms conventional industrial catalysts that use high concentrations of platinum (10,000 ppm).
Propylene is commonly produced through the propane dehydrogenation (PDH) process, which removes hydrogen from propane. Platinum has long been used as a catalyst in PDH due to its high efficiency in breaking carbon-hydrogen bonds and facilitating hydrogen removal. However, platinum is costly and suffers from performance degradation over repeated use.
To address this, the KAIST team engineered a catalyst that incorporates only a minimal amount of platinum, relying on gallium and alumina as the primary components.
< Figure 1. Schematic diagram showing the catalytic cooperation between gallium (Ga) and platinum (Pt) >
The core mechanism of the catalyst involves a cooperative function between the metals: gallium activates the C–H bond in propane to produce propylene, while platinum bonds the residual hydrogen atoms on the surface to form hydrogen gas (H₂), which is then released. This division of roles allows for high catalytic efficiency despite the drastic reduction in platinum content.
The researchers identified an optimal platinum-to-gallium ratio that delivered peak performance and provided a scientific rationale and quantitative metrics to predict this ideal composition.
Additionally, the team addressed a major limitation of traditional platinum catalysts: sintering—the agglomeration of platinum particles during repeated use, which causes performance loss. By adding a small amount of cerium (Ce), the researchers successfully suppressed this aggregation. As a result, the new catalyst maintained stable performance even after more than 20 reaction-regeneration cycles.
< Figure 2. Performance comparison of KAIST's newly developed catalyst (100 ppm platinum) and existing commercial platinum catalyst (10,000 ppm platinum) >
Professor Choi stated, “This research demonstrates the possibility of reducing platinum usage to 1/100th of current levels without compromising, and even enhancing, performance. It presents significant economic and environmental advantages, including lower catalyst costs, extended replacement intervals, and reduced catalyst waste.”
He added, “We are planning to evaluate this technology for large-scale process demonstration and commercialization. If adopted in industry, it could greatly improve the economic viability and efficiency of propylene production.”
The study was led by Professor Minkee Choi as corresponding author, with Ph.D. candidate Susung Lee as the first author. The findings were published in the Journal of the American Chemical Society (JACS), a leading journal in chemistry and chemical engineering, on February 13.※ Paper title: Ideal Bifunctional Catalysis for Propane Dehydrogenation over Pt-Promoted Gallia-Alumina and Minimized Use of Precious Elements※ DOI: https://pubs.acs.org/doi/10.1021/jacs.4c13787
The research was supported by the National Research Foundation of Korea and Hanwha Solutions Corporation.
2025.05.12 View 66 -
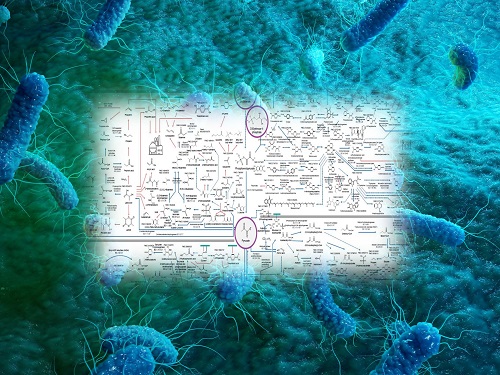 Interactive Map of Metabolical Synthesis of Chemicals
An interactive map that compiled the chemicals produced by biological, chemical and combined reactions has been distributed on the web
- A team led by Distinguished Professor Sang Yup Lee of the Department of Chemical and Biomolecular Engineering, organized and distributed an all-inclusive listing of chemical substances that can be synthesized using microorganisms
- It is expected to be used by researchers around the world as it enables easy assessment of the synthetic pathway through the web.
A research team comprised of Woo Dae Jang, Gi Bae Kim, and Distinguished Professor Sang Yup Lee of the Department of Chemical and Biomolecular Engineering at KAIST reported an interactive metabolic map of bio-based chemicals. Their research paper “An interactive metabolic map of bio-based chemicals” was published online in Trends in Biotechnology on August 10, 2022.
As a response to rapid climate change and environmental pollution, research on the production of petrochemical products using microorganisms is receiving attention as a sustainable alternative to existing methods of productions. In order to synthesize various chemical substances, materials, and fuel using microorganisms, it is necessary to first construct the biosynthetic pathway toward desired product by exploration and discovery and introduce them into microorganisms. In addition, in order to efficiently synthesize various chemical substances, it is sometimes necessary to employ chemical methods along with bioengineering methods using microorganisms at the same time. For the production of non-native chemicals, novel pathways are designed by recruiting enzymes from heterologous sources or employing enzymes designed though rational engineering, directed evolution, or ab initio design.
The research team had completed a map of chemicals which compiled all available pathways of biological and/or chemical reactions that lead to the production of various bio-based chemicals back in 2019 and published the map in Nature Catalysis. The map was distributed in the form of a poster to industries and academia so that the synthesis paths of bio-based chemicals could be checked at a glance.
The research team has expanded the bio-based chemicals map this time in the form of an interactive map on the web so that anyone with internet access can quickly explore efficient paths to synthesize desired products. The web-based map provides interactive visual tools to allow interactive visualization, exploration, and analysis of complex networks of biological and/or chemical reactions toward the desired products. In addition, the reported paper also discusses the production of natural compounds that are used for diverse purposes such as food and medicine, which will help designing novel pathways through similar approaches or by exploiting the promiscuity of enzymes described in the map. The published bio-based chemicals map is also available at http://systemsbiotech.co.kr.
The co-first authors, Dr. Woo Dae Jang and Ph.D. student Gi Bae Kim, said, “We conducted this study to address the demand for updating the previously distributed chemicals map and enhancing its versatility.” “The map is expected to be utilized in a variety of research and in efforts to set strategies and prospects for chemical production incorporating bio and chemical methods that are detailed in the map.”
Distinguished Professor Sang Yup Lee said, “The interactive bio-based chemicals map is expected to help design and optimization of the metabolic pathways for the biosynthesis of target chemicals together with the strategies of chemical conversions, serving as a blueprint for developing further ideas on the production of desired chemicals through biological and/or chemical reactions.”
The interactive metabolic map of bio-based chemicals.
2022.08.11 View 13896
Interactive Map of Metabolical Synthesis of Chemicals
An interactive map that compiled the chemicals produced by biological, chemical and combined reactions has been distributed on the web
- A team led by Distinguished Professor Sang Yup Lee of the Department of Chemical and Biomolecular Engineering, organized and distributed an all-inclusive listing of chemical substances that can be synthesized using microorganisms
- It is expected to be used by researchers around the world as it enables easy assessment of the synthetic pathway through the web.
A research team comprised of Woo Dae Jang, Gi Bae Kim, and Distinguished Professor Sang Yup Lee of the Department of Chemical and Biomolecular Engineering at KAIST reported an interactive metabolic map of bio-based chemicals. Their research paper “An interactive metabolic map of bio-based chemicals” was published online in Trends in Biotechnology on August 10, 2022.
As a response to rapid climate change and environmental pollution, research on the production of petrochemical products using microorganisms is receiving attention as a sustainable alternative to existing methods of productions. In order to synthesize various chemical substances, materials, and fuel using microorganisms, it is necessary to first construct the biosynthetic pathway toward desired product by exploration and discovery and introduce them into microorganisms. In addition, in order to efficiently synthesize various chemical substances, it is sometimes necessary to employ chemical methods along with bioengineering methods using microorganisms at the same time. For the production of non-native chemicals, novel pathways are designed by recruiting enzymes from heterologous sources or employing enzymes designed though rational engineering, directed evolution, or ab initio design.
The research team had completed a map of chemicals which compiled all available pathways of biological and/or chemical reactions that lead to the production of various bio-based chemicals back in 2019 and published the map in Nature Catalysis. The map was distributed in the form of a poster to industries and academia so that the synthesis paths of bio-based chemicals could be checked at a glance.
The research team has expanded the bio-based chemicals map this time in the form of an interactive map on the web so that anyone with internet access can quickly explore efficient paths to synthesize desired products. The web-based map provides interactive visual tools to allow interactive visualization, exploration, and analysis of complex networks of biological and/or chemical reactions toward the desired products. In addition, the reported paper also discusses the production of natural compounds that are used for diverse purposes such as food and medicine, which will help designing novel pathways through similar approaches or by exploiting the promiscuity of enzymes described in the map. The published bio-based chemicals map is also available at http://systemsbiotech.co.kr.
The co-first authors, Dr. Woo Dae Jang and Ph.D. student Gi Bae Kim, said, “We conducted this study to address the demand for updating the previously distributed chemicals map and enhancing its versatility.” “The map is expected to be utilized in a variety of research and in efforts to set strategies and prospects for chemical production incorporating bio and chemical methods that are detailed in the map.”
Distinguished Professor Sang Yup Lee said, “The interactive bio-based chemicals map is expected to help design and optimization of the metabolic pathways for the biosynthesis of target chemicals together with the strategies of chemical conversions, serving as a blueprint for developing further ideas on the production of desired chemicals through biological and/or chemical reactions.”
The interactive metabolic map of bio-based chemicals.
2022.08.11 View 13896 -
 A Comprehensive Metabolic Map for Bio-Based Chemicals Production
A KAIST research team completed a metabolic map that charts all available strategies and pathways of chemical reactions that lead to the production of various industrial bio-based chemicals.
The team was led by Distinguished Professor Sang Yup Lee, who has produced high-quality metabolic engineering and systems engineering research for decades, and made the hallmark chemicals map after seven years of studies.
The team presented a very detailed analysis on metabolic engineering for the production of a wide range of industrial chemicals, fuels, and materials. Surveying the current trends in the bio-based production of chemicals in industrial biotechnology, the team thoroughly examined the current status of industrial chemicals produced using biological and/or chemical reactions.
This comprehensive map is expected to serve as a blueprint for the visual and intuitive inspection of biological and/or chemical reactions for the production of interest from renewable resources. The team also compiled an accompanying poster to visually present the synthetic pathways of chemicals in the context of their microbial metabolism.
As metabolic engineering has become increasing powerful in addressing limited fossil resources, climate change, and other environmental issues, the number of microbially produced chemicals using biomass as a carbon source has increased substantially. The sustainable production of industrial chemicals and materials has been explored with micro-organisms as cell factories and renewable nonfood biomass as raw materials for alternative petroleum. The engineering of these micro-organism has increasingly become more efficient and effective with the help of metabolic engineering – a practice of engineering using the metabolism of living organisms to produce a desired metabolite.
With the establishment of systems metabolic engineering – the integration of metabolic engineering with tools and strategies from systems biology, synthetic biology and evolutionary engineering – the speed at which micro-organisms are being engineered has reached an unparalleled pace.
In order to evaluate the current state at which metabolically engineered micro-organisms can produce a large portfolio of industrial chemicals, the team conducted an extensive review of the literature and mapped them out on a poster. This resulting poster, termed the bio-based chemicals map, presents synthetic pathways for industrial chemicals, which consist of biological and/or chemical reactions.
Industrial chemicals and their production routes are presented in the context of central carbon metabolic pathways as these key metabolites serve as precursors for the chemicals to be produced. The resulting biochemical map allows the detection and analysis of optimal synthetic pathways for a given industrial chemical. In addition to the poster, the authors have compiled a list of chemicals that have successfully been produced using micro-organisms and a list of the corresponding companies producing them commercially. This thorough review of the literature and the accompanying analytical summary will be an important resource for researchers interested in the production of chemicals from renewable biomass sources.
Metabolically engineered micro-organisms have already made a huge contribution toward the sustainable production of chemicals using renewable resources. Professor Lee said he wanted a detailed survey of the current state and capacity of bio-based chemicals production.
“We are so excited that this review and poster will expand further discussion on the production of important chemicals through engineered micro-organisms and also combined biological and chemical means in a more sustainable manner,” he explained.
This work was supported by the Technology Development Program to Solve Climate Changes on Systems Metabolic Engineering for Biofineries from the Ministry of Science and ICT through the National Research Foundation of Korea.
For further information, Distinguished Professor Sang Yup Lee of the Department of Chemical and Biomolecular Engineering at KAIST ( leesy@kaist.ac.kr , Tel: +82-42-350-3930)
Figure: Bio-based chemicals production through biological and chemical routes. This metabolic map describes representative chemicals that can be produced either by biological and/or chemical means. Red arrows represent chemical routes and blue arrows represent biological routes. Intermediate metabolites in the metabolism of a living organism can serve as a platform toward the production of industrially relevant chemicals. A more comprehensive map presented by the team can be found as a poster in the review.
2019.01.15 View 7467
A Comprehensive Metabolic Map for Bio-Based Chemicals Production
A KAIST research team completed a metabolic map that charts all available strategies and pathways of chemical reactions that lead to the production of various industrial bio-based chemicals.
The team was led by Distinguished Professor Sang Yup Lee, who has produced high-quality metabolic engineering and systems engineering research for decades, and made the hallmark chemicals map after seven years of studies.
The team presented a very detailed analysis on metabolic engineering for the production of a wide range of industrial chemicals, fuels, and materials. Surveying the current trends in the bio-based production of chemicals in industrial biotechnology, the team thoroughly examined the current status of industrial chemicals produced using biological and/or chemical reactions.
This comprehensive map is expected to serve as a blueprint for the visual and intuitive inspection of biological and/or chemical reactions for the production of interest from renewable resources. The team also compiled an accompanying poster to visually present the synthetic pathways of chemicals in the context of their microbial metabolism.
As metabolic engineering has become increasing powerful in addressing limited fossil resources, climate change, and other environmental issues, the number of microbially produced chemicals using biomass as a carbon source has increased substantially. The sustainable production of industrial chemicals and materials has been explored with micro-organisms as cell factories and renewable nonfood biomass as raw materials for alternative petroleum. The engineering of these micro-organism has increasingly become more efficient and effective with the help of metabolic engineering – a practice of engineering using the metabolism of living organisms to produce a desired metabolite.
With the establishment of systems metabolic engineering – the integration of metabolic engineering with tools and strategies from systems biology, synthetic biology and evolutionary engineering – the speed at which micro-organisms are being engineered has reached an unparalleled pace.
In order to evaluate the current state at which metabolically engineered micro-organisms can produce a large portfolio of industrial chemicals, the team conducted an extensive review of the literature and mapped them out on a poster. This resulting poster, termed the bio-based chemicals map, presents synthetic pathways for industrial chemicals, which consist of biological and/or chemical reactions.
Industrial chemicals and their production routes are presented in the context of central carbon metabolic pathways as these key metabolites serve as precursors for the chemicals to be produced. The resulting biochemical map allows the detection and analysis of optimal synthetic pathways for a given industrial chemical. In addition to the poster, the authors have compiled a list of chemicals that have successfully been produced using micro-organisms and a list of the corresponding companies producing them commercially. This thorough review of the literature and the accompanying analytical summary will be an important resource for researchers interested in the production of chemicals from renewable biomass sources.
Metabolically engineered micro-organisms have already made a huge contribution toward the sustainable production of chemicals using renewable resources. Professor Lee said he wanted a detailed survey of the current state and capacity of bio-based chemicals production.
“We are so excited that this review and poster will expand further discussion on the production of important chemicals through engineered micro-organisms and also combined biological and chemical means in a more sustainable manner,” he explained.
This work was supported by the Technology Development Program to Solve Climate Changes on Systems Metabolic Engineering for Biofineries from the Ministry of Science and ICT through the National Research Foundation of Korea.
For further information, Distinguished Professor Sang Yup Lee of the Department of Chemical and Biomolecular Engineering at KAIST ( leesy@kaist.ac.kr , Tel: +82-42-350-3930)
Figure: Bio-based chemicals production through biological and chemical routes. This metabolic map describes representative chemicals that can be produced either by biological and/or chemical means. Red arrows represent chemical routes and blue arrows represent biological routes. Intermediate metabolites in the metabolism of a living organism can serve as a platform toward the production of industrially relevant chemicals. A more comprehensive map presented by the team can be found as a poster in the review.
2019.01.15 View 7467 -
 KAIST and Saudi Aramco agreed to establish a joint CO2 research center in Korea
The Korea Advanced Institute of Science and Technology (KAIST) and Saudi Aramco, a global energy and petrochemicals enterprise, signed a memorandum of understanding (MOU) on January 6, 2013 in Dhahran, Saudi Arabia and pledged to jointly collaborate in research and development of innovative technologies and solutions to address the world"s energy challenges.
Under the MOU, the two entities agreed to establish a research center, Saudi Aramco-KAIST CO2 Research Center, near KAIST"s main campus in Daejeon, Korea. The research center, to be jointly managed by KAIST and Saudi Aramco, will foster and facilitate research collaborations in areas such as tackling carbon dioxide (CO2) emissions by removal or capture of CO2, conversing CO2 into useful products, developing efficiency improvements in energy production, sharing carbon management technologies, establishing exchange programs, and conducting joint projects.
According to Saudi Aramco, the company"s collaboration with KAIST is the first partnership established in Asia. Khalid A. Al-Falih, President and CEO of Saudi Aramco, said,
"The CO2 Research Center represents a major step in Saudi Aramco"s research and technology strategy to partner with top global institutions to help address and find sustainable solutions to the world’s energy challenge both domestically and internationally."
2013.03.19 View 10574
KAIST and Saudi Aramco agreed to establish a joint CO2 research center in Korea
The Korea Advanced Institute of Science and Technology (KAIST) and Saudi Aramco, a global energy and petrochemicals enterprise, signed a memorandum of understanding (MOU) on January 6, 2013 in Dhahran, Saudi Arabia and pledged to jointly collaborate in research and development of innovative technologies and solutions to address the world"s energy challenges.
Under the MOU, the two entities agreed to establish a research center, Saudi Aramco-KAIST CO2 Research Center, near KAIST"s main campus in Daejeon, Korea. The research center, to be jointly managed by KAIST and Saudi Aramco, will foster and facilitate research collaborations in areas such as tackling carbon dioxide (CO2) emissions by removal or capture of CO2, conversing CO2 into useful products, developing efficiency improvements in energy production, sharing carbon management technologies, establishing exchange programs, and conducting joint projects.
According to Saudi Aramco, the company"s collaboration with KAIST is the first partnership established in Asia. Khalid A. Al-Falih, President and CEO of Saudi Aramco, said,
"The CO2 Research Center represents a major step in Saudi Aramco"s research and technology strategy to partner with top global institutions to help address and find sustainable solutions to the world’s energy challenge both domestically and internationally."
2013.03.19 View 10574 -
 Industrial Liaison Program Membership Implemented
KAIST implemented, for the first time as a Korean University, the Industrial Liaison Program Membership (ILPM).
ILPM is a structure where it does not limit the university as a minor technological counseling institute and encourages the university to provide expert services that the companies need in a proactive manner.
The ILPM is an Industry-Scholar Cooperative Model that offers companies with patents, technologies, labor force, research tools, and information to the companies all the while serving as the leader in research and development that will bring competitiveness to the company.
The first member of the ILPM at KAIST is ‘Yeul Chon’ Chemicals which is a subsidiary of the Nong-Shim Group and is a leading group in the field of high tech packaging, film and environmentally friendly materials.
KAIST and Yeul Chon Chemicals signed a MOU for technological cooperation and agreement to become a member of ILPM at KAIST on the 22nd of March.
With the agreement, the Yeul Chon Chemicals will now have access to all of KAIST’s information, technology, students, and counseling from professors.
2011.04.13 View 9898
Industrial Liaison Program Membership Implemented
KAIST implemented, for the first time as a Korean University, the Industrial Liaison Program Membership (ILPM).
ILPM is a structure where it does not limit the university as a minor technological counseling institute and encourages the university to provide expert services that the companies need in a proactive manner.
The ILPM is an Industry-Scholar Cooperative Model that offers companies with patents, technologies, labor force, research tools, and information to the companies all the while serving as the leader in research and development that will bring competitiveness to the company.
The first member of the ILPM at KAIST is ‘Yeul Chon’ Chemicals which is a subsidiary of the Nong-Shim Group and is a leading group in the field of high tech packaging, film and environmentally friendly materials.
KAIST and Yeul Chon Chemicals signed a MOU for technological cooperation and agreement to become a member of ILPM at KAIST on the 22nd of March.
With the agreement, the Yeul Chon Chemicals will now have access to all of KAIST’s information, technology, students, and counseling from professors.
2011.04.13 View 9898 -
 Prof. Sang-Yup Lee Receives Merck Award for Metabolic Engineering
Prof. Sang-Yup Lee of KAIST"s Department of Chemical and Biomolecular Engineering has been chosen as the winner of the 2008 Merck Award for Metabol;ic Engineering established by the world"s leading pharmaceutical and chemical company Merck, KAIST officials said Tuesday, Sept. 16.
The Distinguished Professor of KAIST and LG Chem Chair Professor will receive the award on Sept. 18 during the 7th Metabolic Engineering convention now underway at Puerto Vallarta, Mexico. Prof. Lee will give a commemorative lecture, titled "Systems Metabolic Engineering for Chemicals," at the biannual academic conference. Prof. Lee is the fourth to win the coveted award which is given to the world"s top expert in metabolic engineering with outstanding achievements in the field.
Prof. Lee, 44, who graduated from Seoul National University and earned his master"s and doctoral degrees in chemical engineering from Northwestern University of the United States, is now the dean of the College of Life Science and Bioengineering, KAIST. Since 1994, he has served as the head of the Metabolic and Biomolecular Engineering National Research Laboratory, director of the BioProcess Engineering Center, Director of the Bioinformatics Research Center and Co-Director of the Institute for the BioCentury in KAIST.
Prof. Lee said he was receiving the Merck award "as a representative of KAIST graduates, students and researchers" who have worked with him at the Metabolic Engineering Lab. He added he was happy to see the outcome of bioengineering development projects supported by the Ministry of Education, Science and Technology over the past years was now being recognized by the world"s leading scientific society with the Merck Award.
Metabolic engineering, the art of optimizing genetic and regulatory processes within cells to increase the cell"s production of a certain substance, develops technologies that hold the key to the resolution of the world"s energy, food and environmental problems. The indispensible technology in bioengineering can be applied to the production of biomass to obtain alternative fuel.
Prof. Lee has actively participated in publishing such academic periodicals as Biotechnology Journal (as chief editor), Biotechnology and Bioengineering (deputy editor) and Metabolic Engineering (a member of the editorial committee).
2008.09.17 View 16019
Prof. Sang-Yup Lee Receives Merck Award for Metabolic Engineering
Prof. Sang-Yup Lee of KAIST"s Department of Chemical and Biomolecular Engineering has been chosen as the winner of the 2008 Merck Award for Metabol;ic Engineering established by the world"s leading pharmaceutical and chemical company Merck, KAIST officials said Tuesday, Sept. 16.
The Distinguished Professor of KAIST and LG Chem Chair Professor will receive the award on Sept. 18 during the 7th Metabolic Engineering convention now underway at Puerto Vallarta, Mexico. Prof. Lee will give a commemorative lecture, titled "Systems Metabolic Engineering for Chemicals," at the biannual academic conference. Prof. Lee is the fourth to win the coveted award which is given to the world"s top expert in metabolic engineering with outstanding achievements in the field.
Prof. Lee, 44, who graduated from Seoul National University and earned his master"s and doctoral degrees in chemical engineering from Northwestern University of the United States, is now the dean of the College of Life Science and Bioengineering, KAIST. Since 1994, he has served as the head of the Metabolic and Biomolecular Engineering National Research Laboratory, director of the BioProcess Engineering Center, Director of the Bioinformatics Research Center and Co-Director of the Institute for the BioCentury in KAIST.
Prof. Lee said he was receiving the Merck award "as a representative of KAIST graduates, students and researchers" who have worked with him at the Metabolic Engineering Lab. He added he was happy to see the outcome of bioengineering development projects supported by the Ministry of Education, Science and Technology over the past years was now being recognized by the world"s leading scientific society with the Merck Award.
Metabolic engineering, the art of optimizing genetic and regulatory processes within cells to increase the cell"s production of a certain substance, develops technologies that hold the key to the resolution of the world"s energy, food and environmental problems. The indispensible technology in bioengineering can be applied to the production of biomass to obtain alternative fuel.
Prof. Lee has actively participated in publishing such academic periodicals as Biotechnology Journal (as chief editor), Biotechnology and Bioengineering (deputy editor) and Metabolic Engineering (a member of the editorial committee).
2008.09.17 View 16019